Practical Guidance for Patients Before You Proceed
Before deciding on stem cell therapy for knee pain or osteoarthritis, it’s important for
patients to take a thoughtful, well-informed approach. Regenerative medicine offers potential benefits,
but it should always be considered within the framework of scientific evidence and ethical medical practice.
Begin by confirming that conservative treatments such as physiotherapy, exercise, weight management, and medication have been fully optimized. Many individuals can achieve satisfactory relief through these established methods, which remain the first line of care recommended by orthopedic specialists.
When considering a stem cell-based procedure, ensure that your healthcare provider clearly explains the level of current scientific evidence, expected outcomes, and any potential limitations. Transparent communication about treatment alternatives—such as platelet-rich plasma (PRP) therapy, hyaluronic acid injections, or surgical options—helps patients make balanced, realistic decisions.
Patients are strongly encouraged to seek treatment within a properly regulated clinical environment— preferably an accredited medical centre or research-based trial that documents its outcomes and follows international ethical standards. Avoid unverified clinics or commercial setups that make unsubstantiated promises of “permanent cures.”
Finally, discuss all financial and procedural details before beginning treatment. A trustworthy
centre will provide a clear cost structure, explain the follow-up schedule, and outline options in case
the therapy does not yield the expected benefit. Informed consent and patient autonomy are key pillars of responsible regenerative care.
At Viezec, every patient receives honest medical guidance, transparent information, and professional care delivered under approved clinical standards—ensuring both safety and trust throughout the treatment journey.



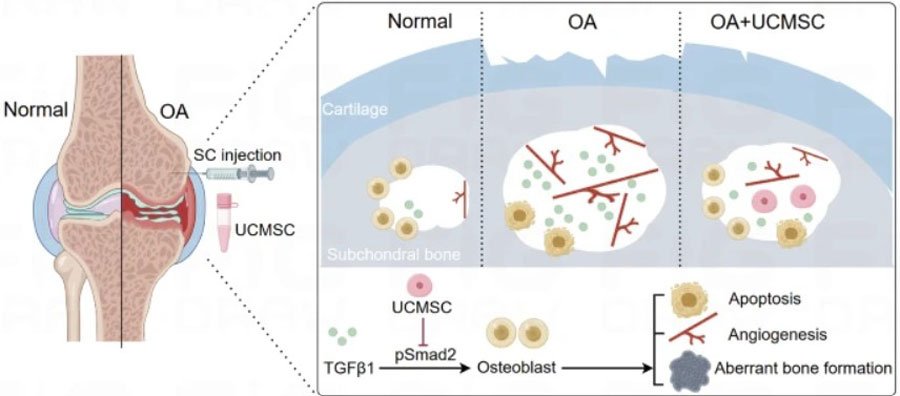 Illustration: targeted intra-articular delivery and paracrine activity of MSCs.
Illustration: targeted intra-articular delivery and paracrine activity of MSCs.

