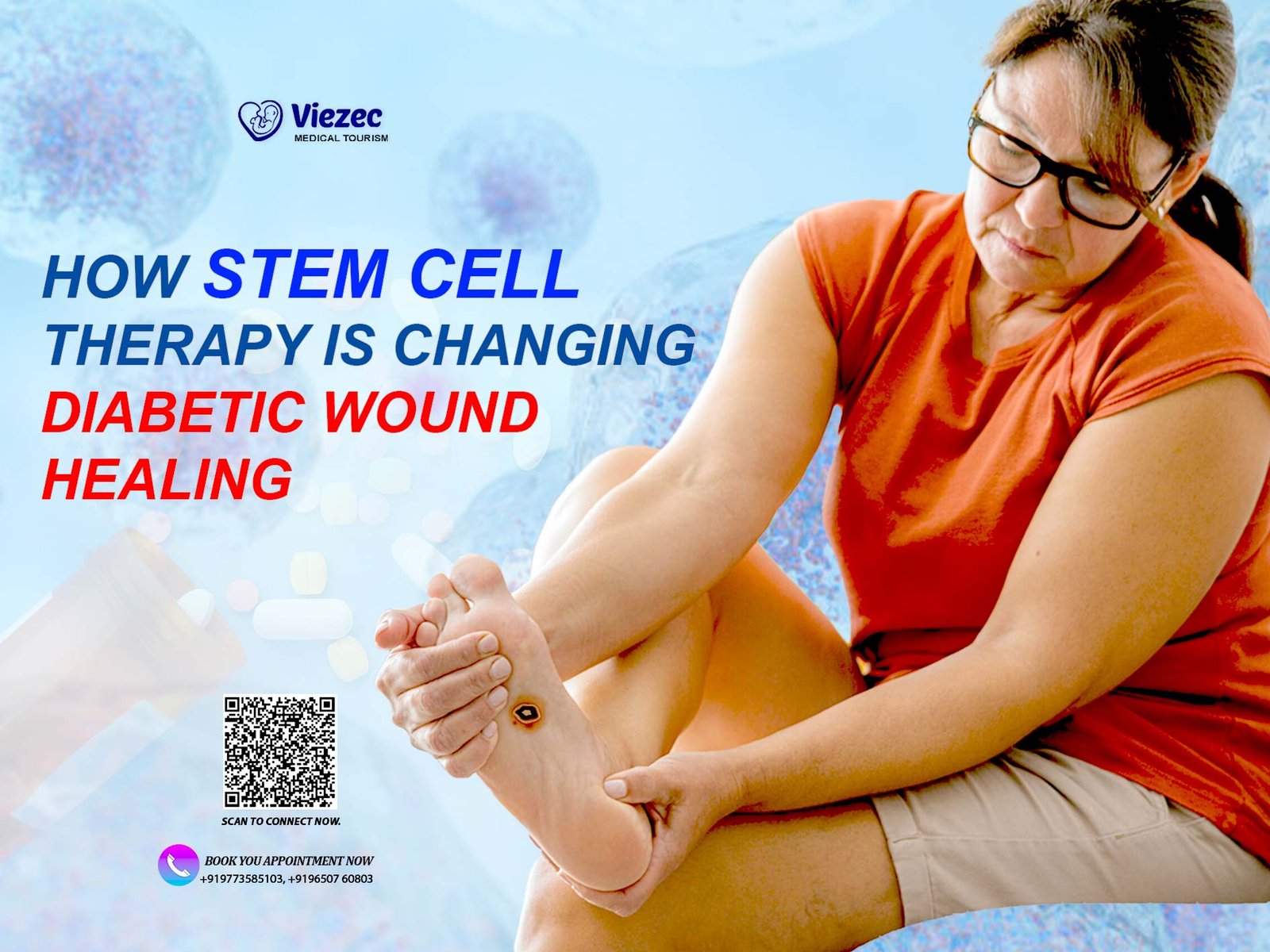
In the rapidly advancing field of regenerative medicine, induced pluripotent stem cells (iPSCs) have emerged as a groundbreaking technology with immense potential. iPSCs represent a significant leap forward in our ability to manipulate cellular behavior and hold promise for revolutionary advancements in disease modeling, drug discovery, and personalized medicine. This article delves into the world of induced pluripotent stem cells, exploring their origin, characteristics, applications, challenges, and the ethical considerations surrounding their use.
Background
The journey into the realm of induced pluripotent stem cells began with the groundbreaking discovery by Shinya Yamanaka and Kazutoshi Takahashi in 2006. Yamanaka and Takahashi demonstrated that adult somatic cells could be reprogrammed into a pluripotent state, resembling embryonic stem cells, by introducing specific transcription factors. This discovery marked a turning point in stem cell research, as it opened new avenues for generating patient-specific cells without the ethical concerns associated with embryonic stem cells.
Understanding Induced Pluripotent Stem Cells
What are Induced Pluripotent Stem Cells?
Induced pluripotent stem cells are artificially created stem cells that possess the unique ability to differentiate into various cell types found in the human body. Unlike embryonic stem cells, which are derived from embryos, iPSCs are generated by reprogramming adult somatic cells, such as skin cells or blood cells. This reprogramming involves the introduction of specific transcription factors, known as Yamanaka factors, which induce a pluripotent state in the target cells.
Yamanaka Factors
The Yamanaka factors, named after Nobel laureate Shinya Yamanaka, consist of a set of transcription factors critical for inducing pluripotency in somatic cells. These factors include Oct4, Sox2, Klf4, and c-Myc. Their concerted action triggers a cascade of molecular events that lead to the erasure of cell identity and the acquisition of pluripotent characteristics. Understanding the role and regulation of these factors is crucial for successful iPSC generation.
Characteristics of Induced Pluripotent Stem Cells
Pluripotency
The hallmark feature of induced pluripotent stem cells is their pluripotency, which means they have the potential to differentiate into any cell type in the human body. This unique property makes iPSCs a versatile tool for regenerative medicine, as they can be directed to become specific cell types needed for therapeutic applications.
Self-Renewal
Another key characteristic of iPSCs is their ability to self-renew indefinitely. This self-renewal capacity ensures a stable and abundant source of pluripotent cells for research and therapeutic purposes. Understanding the molecular mechanisms governing self-renewal is essential for harnessing the full potential of iPSCs.
Genetic Stability
Maintaining genetic stability is crucial for the safe and effective use of induced pluripotent stem cells. Researchers must address challenges related to genetic mutations and epigenetic changes that may occur during the reprogramming process. Ongoing research aims to enhance the genetic integrity of iPSCs to minimize the risk of tumorigenesis and other undesirable outcomes.
Applications of Induced Pluripotent Stem Cells
Disease Modeling
One of the most significant applications of iPSCs is in disease modeling. Researchers can generate iPSCs from patients with specific genetic disorders, allowing them to study the disease’s mechanisms and progression in a controlled laboratory setting. This personalized approach provides valuable insights into disease pathology and facilitates the development of targeted therapies.
Drug Discovery
The pharmaceutical industry has embraced the use of induced pluripotent stem cells for drug discovery and development. iPSCs enable the creation of disease-specific cell models, allowing researchers to screen potential drug candidates more efficiently. This personalized medicine approach holds the promise of identifying safer and more effective drugs tailored to individual patient profiles.
Regenerative Medicine
Induced pluripotent stem cells have transformative implications for regenerative medicine. These cells can be differentiated into various cell types, such as neurons, cardiomyocytes, and pancreatic cells, offering potential treatments for conditions ranging from neurodegenerative disorders to heart disease and diabetes. The ability to replace damaged or diseased tissues with patient-specific cells holds great therapeutic potential.
Challenges and Considerations
Tumorigenicity
One of the major challenges associated with induced pluripotent stem cells is the risk of tumorigenicity. The reprogramming process and prolonged in vitro culture may lead to the formation of teratomas, which are tumors containing cells from all three germ layers. Addressing this challenge requires refining reprogramming techniques and developing stringent quality control measures to ensure the safety of iPSC-derived therapies.
Epigenetic Memory
Another consideration in iPSC research is the presence of epigenetic memory from the donor cells. The original cell type’s epigenetic marks may persist in the iPSCs, influencing their differentiation potential. Understanding and mitigating epigenetic memory are essential for ensuring the faithful differentiation of iPSCs into desired cell types.
Ethical Considerations
Ethical Advantages
The use of induced pluripotent stem cells carries ethical advantages compared to embryonic stem cells. By avoiding the use of embryos, iPSC research sidesteps ethical concerns related to embryo destruction and the potential for exploitation. This ethical advantage has contributed to the broader acceptance and support for iPSC research.
Ethical Challenges
While iPSCs offer ethical advantages, ethical challenges persist. Issues related to informed consent, privacy, and the potential commodification of cells must be carefully addressed. Establishing clear ethical guidelines and governance structures is crucial to navigating these challenges responsibly.
Future Directions and Innovations
CRISPR/Cas9 Technology
Integration of CRISPR/Cas9 technology with induced pluripotent stem cells opens new avenues for precise genome editing. This powerful tool allows researchers to correct genetic mutations, study gene function, and enhance the therapeutic potential of iPSCs. The synergy between CRISPR/Cas9 and iPSC technology holds great promise for advancing precision medicine.
3D Bioprinting
The field of 3D bioprinting is revolutionizing tissue engineering and regenerative medicine. Combining induced pluripotent stem cells with 3D bioprinting technologies enables the creation of complex, functional tissues. This innovative approach holds potential for producing transplantable organs and tissues tailored to individual patient needs.
Induced pluripotent stem cells represent a paradigm shift in the field of regenerative medicine. Their ability to be derived from adult cells, coupled with their pluripotency, opens new possibilities for personalized medicine, disease modeling, and drug discovery. As researchers continue to overcome challenges and harness the full potential of iPSCs, the future holds exciting prospects for transforming healthcare and improving patient outcomes.
In conclusion, the journey of induced pluripotent stem cells from discovery to application underscores the dynamic nature of scientific progress. With ongoing research, innovations, and ethical considerations, iPSCs are poised to play a pivotal role in shaping the future of medicine and biology.